G2013
Western Bean Cutworm in Corn and Dry Beans
This NebGuide addresses the life cycle, scouting and treatment of the western bean cutworm in corn and dry beans.
Ronald C. Seymour, Extension Educator
Gary L. Hein, Professor of Entomology
Robert J. Wright, Extension Entomologist
- Life History
- Infestations on Corn
- Infestations on Dry Beans
- Sampling Corn
- Sampling Dry Beans
- Pest Management
Western bean cutworm (WBC) can be a severe pest in corn and dry beans. Larval feeding damages both crops through reduced yield and quality. In corn, direct feeding losses may be compounded by fungal infections associated with larval feeding and waste products. In dry beans, damaged or “worm-chewed” beans are a significant quality factor for both processed and bagged dry beans. Western bean cutworm infestations occur every year in western Nebraska and the surrounding region, but can be found in high numbers throughout the state. Traditionally, the western bean cutworm has largely been limited to areas in the western Great Plains, but in the last 10 years, western bean cutworm has steadily spread eastward through the Corn Belt to as far east as Pennsylvania.
Life History
Western bean cutworm has one generation per year with moth emergence usually beginning in early July. The emergence date can be predicted by calculating growing degree days. Starting heat unit accumulations on May 1, using a base air temperature of 50°F, growing degree days for 25 percent, 50 percent, and 75 percent moth emergence are 1319, 1422, and 1536, respectively.
Populations vary from year to year, but there is a tendency for greater populations to occur every six to eight years. Throughout the western Great Plains region, western bean cutworm populations are greater in fields with sandy soils.
Western bean cutworm moths are about 3/4 inch long with a wing span of about 1 1/2 inches (Figure 1). The body is light brown, and the wings are generally dark brown with a distinctive pattern. The front wings have a broad white or cream stripe that runs two-thirds of the length of the leading edge. Behind this stripe is a central white spot and, further away from the body, a half moon shaped spot. The hind wings are cream colored without markings. When at rest, the wings are swept back over the body. The moths are strong fliers and are known to travel several miles. Female moths emit a pheromone (scent) that attracts males for mating.
After mating, eggs are usually laid on either field corn, popcorn, sweet corn, or dry beans. Tomatoes and fruits of nightshade and ground cherry are acceptable but non-preferred hosts. Eggs are laid in masses of 5 to 200 with an average of about 50 eggs per mass (Figure 2). The eggs are 0.03 inches in diameter, dome shaped with ridges and reticulations. When first laid, the eggs are pearly white, but within two days they turn tan. Egg development usually takes five to seven days and the eggs turn dark purple shortly (less than 24 hours) before hatching (Figure 3).
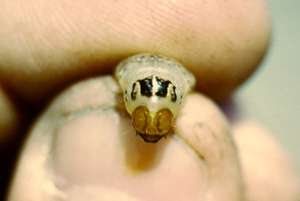
After egg hatch, the larvae remain clumped near the egg mass for several hours, feeding on the chorion (shells) of the eggs. The larvae then move to protected feeding sites, the location depending on the growth stage of the host. Larvae feed for about 31 days and develop through five stages (instars) on the host plant. First instar larvae are quite mobile and may infest several adjacent plants. They are dark brown with faint crosshatched markings on their backs (Figure 4). As the larvae develop, they become light tan to pink and the crosshatch markings on their backs become more distinct (Figure 5). Third instar and older western bean cutworm larvae also can be differentiated from other cutworms and caterpillars feeding on the host plant by three characteristic dark brown stripes immediately behind the head (Figure 6). Larvae continue to feed through the fifth instar after which they drop to the ground, burrow 3 to 6 inches into the soil, and construct an earthen overwintering cell (Figure 7). They spend the winter inside this cell in a pre-pupal stage. Larvae pupate in late May followed by adult emergence starting in early July.
 |
 |
|||
| Figure 1. Adult western bean cutworm. | Figure 2. Newly laid western bean cutworm eggs. | |||
 |
 |
|||
| Figure 3. Eggs immediately prior to hatch. | Figure 4. Newly hatched larvae. | |||
 |
 |
|||
| Figure 5. Larger larva on corn ear. | Figure 6. Larva with stripes on prothorax. | |||
|
||||
Infestations on Corn
Western bean cutworm females often enter the whorl of the plant where they lay eggs on the upper surface of corn leaves. Fields still in the whorl stage are preferred for oviposition (egg-laying). Most eggs hatch, but only a small percentage of the larvae actually survive to maturity. In pre-tassel corn, newly hatched larvae move to the whorl where they feed on the flag leaf, the flowers of the tassel, and other yellow tissue (Figure 8). Once tasseling begins, newly hatched larvae feed within the tassel, leaf axils, or the green silks of the developing ear. Once pollen shed is complete and the tassels dry up, the larvae move to the silks. Through this dispersal behavior, larvae from one egg mass may infest several plants down the row and in immediately adjacent rows in an area 6 to 10 feet in diameter. Second and third instar larvae continue to feed on the silks and move into the ear to feed on the kernels of the developing ears. Fourth instar larvae feed primarily on kernels near the ear tip (Figure 5). If the ear tips are crowded, some larvae may move to the outside of the ear, chew through the husks, and initiate feeding on the kernels (Figure 9).
 |
 |
|
| Figure 8. Larvae in corn tassel. | Figure 9. Cutworm damaged corn ears. |
Research has shown that a field average of one larva per plant at dent stage can reduce yield by 3.7 bu/ac. Western bean cutworm larvae are not cannibalistic, thus often infestations of multiple larvae per ear may occur. In years with severe infestations, two or more larvae per ear may be common and, although unusual, corn ears infested with 10 or more larvae have been recorded. In these crowded situations, as much as 50 to 60 percent of an ear’s kernels may sustain feeding damage.
Infestations on Dry Beans
Western bean cutworm eggs are laid on the lower surface of bean leaves within the dense canopy of foliage. First instar larvae may disperse up to 12 feet along a row and 10 feet across rows. Larvae remain on the leaves until they are about 1/2 inch long. They feed at night on young leaf material and blossoms. As the larvae grow and ponds develop, they begin to feed in the pods and on the developing seeds (Figure 10). Larger larvae leave the pods during the day, seeking protection in the soil. If the larvae have not completed development when the beans are cut, they may congregate under the windrow and feed on the pods and seeds until harvest (Figure 11). Economic injury levels for yield loss are difficult to determine because of the uncertain impacts on product quality and market price, but the economic injury level for dry beans is about 1 percent damaged seed in the marketed product. This damage level would result from about 4 to 6 percent damaged pods in the field.
 |
 |
|
| Figure 10. Cutworm damage to bean pod. | Figure 11. Cutworm-damaged dry beans. |
Sampling Corn
Western bean cutworm moths can be detected with black light or pheromone traps. Based on light trap catches, most of the eggs are laid during the peak moth flight in mid to late July. Light traps should be monitored regularly until after the adult population peaks. Field scouting should be initiated when western bean cutworm moths are first noticed. The upper surface of the upper leaves of corn plants should be examined for egg masses and/or small larvae. Before pollen shed, the tassels also should be inspected for small larvae. When scouting for western bean cutworms, check 10 consecutive plants in several randomly selected locations of each field. These locations should be representative of all areas of a field. Egg laying also will vary with plant growth stage; therefore, portions of a field planted to hybrids with different maturities should be sampled separately. Checking as few as five locations in the field may be adequate to establish the infestation level, particularly if either a low (0 in 10) or a high (more than 2 in 10) number of plants in each location have egg masses or larvae. If the number of infested plants falls between these extremes, observations in 10 to 15 locations may be required to accurately establish the western bean cutworm damage potential for the field.
If 5 to 8 percent of field corn plants have egg masses and/or small larvae, consider an insecticide application. This threshold or infestation level may need to be adjusted based on the crop’s value and control costs. Lower crop values and higher insecticide costs would suggest use of a higher economic threshold value. If an insecticide application is required, timing is critical. If the eggs have hatched, insecticide applications should be made after 95 percent of the plant tassels have emerged, but before the larvae have a chance to enter the silks. Once larvae have moved into the silks and ear tip to feed, insecticide control is more difficult. If the eggs have not hatched and plants have tasseled, the application should be timed for when most of the eggs are expected to hatch. Purple eggs should hatch within about 24 hours.
Sampling Dry Beans
|
Dry beans cannot be effectively scouted for western bean cutworm eggs or small larvae; therefore, it is necessary to use less direct methods to establish a field’s damage potential. Pheromone trap catches may be used to provide an estimate of infestation potential, proper timing of field scouting activities, and optimum timing for initiation of control methods. Inexpensive and effective pheromone traps may be constructed from a one-gallon plastic milk jug (Figure 12) and pheromone may be purchased from a commercial supplier (Table I). Traps are constructed by cutting out the side panels of the jug, leaving a 2-inch bottom reservoir to be filled with a 4:1 mixture of water and antifreeze and a couple drops of dish soap. Moths become trapped in this liquid and can be counted. Pheromone lures may be secured with a pin to the undersurface of the milk jug cap. Scentry™ pheromone lures are recommended because of the variability seen with other brands.
Traps should be mounted at a 4-foot height on posts in two locations at the edge of the bean field. If possible, install traps near lush vegetation, such as a growing corn or sugarbeet field. It also may help to place the traps in the northwest and southeast corners of the field. These steps will ensure that moths will not avoid the traps due to lack of vegetation and the prevailing winds will spread the pheromone over the field, increasing the chance of drawing moths from the field being monitored.
Pheromone traps should be set out in early July. Moths captured in each trap should be counted regularly and the total accumulated over time until the moth flight peaks. During the moth flight, the traps should be emptied and moths counted at least every third day. Longer trapping periods may be acceptable during periods of minimal activity, but in years with high moth counts the traps can quickly exceed their capacity and may need to be counted daily. To ensure optimum moth capture, a fresh antifreeze mixture should be added each time the traps are counted. The date of the peak moth flight should be recorded and the cumulative number of moths, caught from the initiation of the flight until the peak, should be calculated. If the cumulative catch at the peak of the moth flight is less than 700 per trap, the risk of significant damage is low. If the number is between 700 and 1,000 moths per trap, the risk of damage is moderate and additional sampling information will be needed to reach a decision. If the total moth count exceeds 1,000 per trap, the risk for damage is high. However, not all high-risk fields will develop economically threatening damage, so additional information will be helpful in reaching a treatment decision. If an insecticide treatment is required, the application should be made 10 to 21 days after the peak moth flight.
|
Treatment decisions often require further information to better establish damage potential of higher risk fields. Significant cutworm infestations in nearby corn fields may signal a potentially damaging population in the neighboring dry beans. Additional information on damage potential can be gained by checking bean pods for feeding damage about three weeks after the peak moth flight. At this time, pod feeding by the partially grown larvae will just be starting. If pod feeding is noticeable (0.5 to 1 percent or more pod damage), an insecticide application should be considered and, if necessary, administered quickly to prevent further damage.
Pest Management
Few cultural methods effectively control western bean cutworms. Disturbing the soil by plowing or disking is thought to reduce overwintering larval survival; however, the effectiveness of this practice on a broad scale has not been tested. A few dry bean varieties have some resistance to feeding damage but the agronomic characteristics of these varieties are not favorable for commercial production.
Early instar western bean cutworms are exposed on the plant and vulnerable to predators. Thus, there are several predators that help reduce western bean cutworm infestations. Nabids, ladybird beetle adults, spiders and perhaps other predators feed on both eggs and larvae up to the third instar. After the third instar of larval development, predation by birds can be beneficial. Blackbirds can cause high levels of mortality on western bean cutworm larvae found in the ear tips of corn plants, especially when the majority of ears are infested with cutworms. In addition to these natural enemies, western bean cutworm larvae are susceptible to a naturally occurring disease caused by the microsporidian, Nosema sp. Although these naturally occurring control methods are important in reducing western bean cutworm infestations, outbreaks that can cause economic loss in corn and dry beans are still common and may require insecticide applications for adequate control.
Insecticide controls target the larvae stage of western bean cutworms. Liquid insecticides applied by airplane or through a center pivot irrigation system typically provide acceptable control. Treatment in corn should target early instar larvae that are still active on the plant because effectiveness decreases as larvae mature and move into the ear to feed. There is some evidence that synthetic pyrethroid insecticides will force larvae out of protective areas due to the irritation properties of the active ingredient. These insecticides may be more effective should the larvae reach the silks prior to treatment. Foliar insecticides that contain Bacillus thuringiensis as the active ingredient do not control western bean cutworms. Current insecticide recommendations are available from your local Extension office or on the University of Nebraska–Lincoln Department of Entomology website at entomology.unl.edu.
The incidence of spider mites in a field should be considered when choosing an insecticide. Some synthetic pyrethroid insecticides may result in an increase (flare) of spider mite infestations. These products do not control the mites but will increase their dispersal within the canopy. Synthetic pyrethroids are also highly toxic to the natural enemies of spider mites, eliminating the population-regulating effect of these beneficial organisms. If spider mites are present and a synthetic pyrethroid is to be applied, the field should be sampled for the potential development of mite infestation and/or a miticide should be considered to be included in the spray mixture.
Many transgenic corn varieties that express toxin(s) produced by B. thuringiensis (Bt corn) are not effective against western bean cutworm. Only the Bt corn hybrids containing the Cry1F toxin (e.g., Herculex I, Herculex XTRA, SmartStax) are labeled for control of western bean cutworm larvae. Control with this transgenic product has been shown to be about 80 percent effective in reducing cutworm numbers.
This publication has been peer reviewed.
Disclaimer Reference to commercial products or trade names is made with the understanding that no discrimination is intended of those not mentioned and no endorsement by University of Nebraska–Lincoln Extension is implied for those mentioned. |
Visit the University of Nebraska–Lincoln Extension Publications Web site for more publications.
Index: Insects and Pests
Field Crops
Issued April 2010

