G1786
White Mold of Dry Beans
White mold symptoms, infection, and control in dry beans for western Nebraska and Colorado.
Robert M. Harveson, Extension Plant Pathologist, Panhandle Research and Extension Center
James R. Steadman, Professor, Plant Pathology
Howard F. Schwartz, Extension Plant Pathologist, Colorado State University
- Introduction
- Signs and Symptoms
- Factors Favoring Disease
- Survival and Infection
- Disease Management
- Fungicide Decision V4 Worksheet for Managing White Mold in Dry Beans
Introduction
One of the most important diseases affecting dry beans in western Nebraska and Colorado is white mold caused by Sclerotinia sclerotiorum. White mold incidence and severity can be sporadic from year to year, but in recent years average losses of 20 percent have been documented, with losses in a few fields exceeding 65 percent.
Signs and Symptoms
White mold is initially observed in fields after flowering with wilting in plant canopies (Figure 1). As the disease progresses, water-soaked spots or lesions form on infected leaves, stems, branches, and pods. These ultimately enlarge into a watery rotten mass of tissue that becomes covered by a white fungal growth (Figure 2). Infection of stems and branches causes affected plant parts to die, taking on a dried bleached appearance (Figure 3). This bleaching symptom is characteristic of pinto and great northern types infected with white mold and differs from the normal tan color resulting from senescence or other diseases. Black and irregularly shaped sclerotia (resting structures of the fungus) form on and within infected plant parts, which also tend to become shredded.
Factors Favoring Disease
Development and spread of white mold is greatly influenced by prevailing environmental conditions and certain agronomic practices. The disease tends to be concentrated in localized areas of the field and only rarely affects an entire field. The disease may still cause substantial yield losses during wet, cool weather near the end of the growing season. High humidity, and wet plant canopies and soil surfaces are necessary for the fungus to spread. Thus, favorable conditions for disease development often occur at the end of the season when temperatures are cooler and foliar growth is dense. Yield loss occurs from rotted pods and reduced seed size on plants with girdled stems and branches (Figure 4). Agronomic practices such as irrigation management, plant density, and cultivar growth habits are all closely linked with the pathogen life cycle (Figure 5). Integrating the management of these cultural factors with chemical spray treatments can effectively reduce disease severity and minimize yield losses to bean crops.
 |
 |
|
Figure 1. Initial foliar wilting symptoms from white mold infection |
Figure 2. White moldy growth on pods and stems, characteristic of advanced white mold infections |
|
 |
 |
|
| Figure 3. White mold-infected plants exhibiting bleached stem and branches | Figure 4. Seeds on the left were harvested from white mold diseased plants, while those on the right came from healthy great northern bean plants |
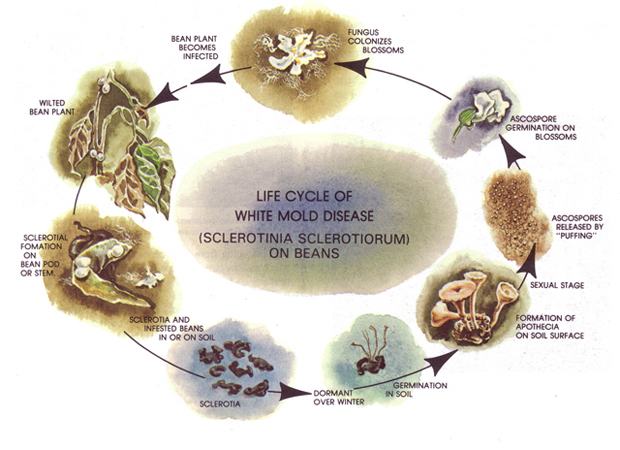 |
Figure 5. Life cycle of white mold disease caused by Sclerotinia Sclerotiorum in dry beans |
Survival and Infection
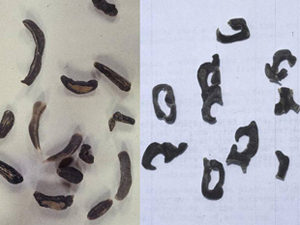
The most important mechanism for pathogen survival is the formation of sclerotia (compact masses of hardened mycelium) that contain reserve food materials. These structures (Figure 6) are produced in abundance in infected plant tissues and reside in soil as infected residues decompose, allowing the fungus to survive during periods of unfavorable environmental conditions. Sclerotia are usually dark-colored and circular to irregular in shape (Figure 7). They range in size from less than ⅛ inch in diameter to the size of a large bean seed. Studies have demonstrated that up to 75 percent of sclerotia buried to a depth of 12 inches will remain alive and infective for at least three years.
S. sclerotiorum also can survive from one season to the next as mycelium in infected bean straw and seeds that have been scattered on the ground during harvest. Post-harvest tillage reduces disease potential for future crops since diseased tissues will be buried and usually decompose in soil before the next bean-growing season. However, sclerotial survival can still pose a problem in subsequent seasons. The pathogen also can be transported within infected seeds or in sclerotia-contaminated seedlots to be planted the next season. This is why it’s important to plant only certified seed that has had sclerotia and poor quality seeds removed during threshing and cleaning operations.
As sclerotia provide the main source of initial inoculum, they must germinate for additional infections to occur. A sclerotium may germinate in one of two ways. The first occurs directly by producing white mycelial strands that seldom exceed ¼ inch in length. These strands do not penetrate living tissue until after dead or senescing tissues such as flowers or leaves have been colonized. This form of germination and infection does not occur frequently in dry bean fields in western Nebraska or Colorado.
More commonly, a sclerotium germinates by producing one or more germ tubes which grow upward from a soil depth of less than 2 inches. Upon reaching the surface, the tip of the germ tube is stimulated to develop into a small (⅛ to ¼ inch diameter) flesh-colored, mushroom-like structure called an apothecium (Figure 8). These structures are first observed 60 days after planting, which coincides with partial to complete row closure. An individual sclerotium can produce numerous apothecia simultaneously or sequentially throughout the growing season, and can produce millions of spores beneath the plant canopy.
Infection resulting from spore dissemination from field to field is not of great importance. Irrigation runoff water is known to transport suspended spores, mycelia, and sclerotia within and between fields. Bumblebees also have been shown to carry spores or other infective fungal structures within or between bean fields.
Like the mycelial strands from directly germinated sclerotia, the spores produced by apothecia must contact and colonize dead or senescing plant tissue on the soil surface within plant canopies (Figure 9). These dead tissues also may be found lodged in branch axils or stuck to newly developing pods. After colonizing the dead tissues, the fungus can directly invade healthy tissue and produce the typical watery soft rot symptoms and white moldy mycelial growth and form new sclerotia, completing the disease cycle. Disease may then progress rapidly within and between adjacent plants. Infection is often localized initially within the more moist and cool irrigation furrows and in low-lying areas of the field.
 |
 |
|
Figure 6. White mold-infected plant with sclerotia embedded in stem and pods |
Figure 7. Sclerotia of varying shapes and sizes, ranging from ⅛ to ½ inch |
|
 |
 |
|
Figure 8. White mold apothecia (approximately ¼ inch in diameter) germinating from sclerotia in the lab |
Figure 9. White mold-colonized bean tissues on soil surface beneath plant canopy |
Disease Management
No single management method effectively prevents the infection process from occurring; however, research has indicated that several measures can help reduce damage from white mold, thereby minimizing yield losses.
- Rotation and tillage practices can be beneficial for reducing the number of sclerotia within fields and their ability to germinate and form spore-producing apothecia, and hence the potential for yield loss.
- Cultural practices, such as over-fertilization, that minimize excessive vine growth also would be beneficial. Thick dense plant growth causes cooler temperatures and more humid conditions within canopies, thus favoring white mold development.
- High plant populations and narrower row widths also may create canopy conditions favorable to white mold development, particularly in fields with a history of the disease.
- Efficient use of irrigation water also is important since disease development depends on there being moist soil beneath the canopy. Infection can be greatly reduced by keeping the soil surface as dry as possible during pod fill and maturation.
- Fungicidal sprays also may be effective for disease management. (See fungicide decision worksheet.) Benomyl (Benlate; no longer labeled for use on beans), thiophanate-methyl (Topsin-M), and iprodione (Rovral) have provided satisfactory management of white mold. Several newer products are available today including boscalid (Endura), prothioconazole (Proline), penthiopyrad (Fontelis, Vertisan); and combination fungicides Priaxor, containing both strobilurin (pyraclostrobin) and carboxamide (fluxapyroxad) components, and Switch containing both cyprodimil (pyrimidine) and fludioxonil (phenylpyrrole). Applications should be made when 80-100 percent of plants have one or more flowers and small pods.
- Timing of fungicide applications is critical to protect as many blossoms as possible from infection. Fungicide must be applied in a sufficient volume of water to provide thorough coverage of all susceptible plant parts. A fungicide application will be more effective for plants with more upright growth habits.
Varietal improvement by breeding for disease resistance and a more open canopy continues to be studied by Carlos Urrea and Mark Brick, bean breeders at the University of Nebraska–Lincoln and Colorado State University, respectively. Their work has substantially improved yields in new releases, but one major issue remains in managing this disease — breaking the positive relationship between high yield and high white mold potential.
Incorporating new bean varieties with an integrated disease management strategy that includes crop rotation, cultural practice modifications, prudent irrigation practices, and the timing and application of proper chemicals will effectively reduce dry bean losses from white mold.
Fungicide Decision V4 Worksheet for Managing White Mold in Dry Beans
Risk Assessment for Common Bean |
YOUR |
|||
| Weekly Total Rainfall, cumulative weekly up to V4 growth stage | ||||
< 0.1 in [ 1 ] |
0.1-0.5 in [ 2 ] |
0.5-1.0 in [ 3 ] |
> 1.0 in [ 4 ] |
|
| Daily High Temperature, average on weekly basis up to V4 stage | ||||
< 64°F [ 4 ] |
64-70°C [ 3 ] |
–82°C [ 2 ] |
< 82°C [ 1 ] |
|
| Humidity (%) or Hours of Dew or Free Water on Foliage, average on weekly basis | ||||
0-25% (< 6 hrs) [ 1 ] |
26-50% (< 12 hrs) [ 2 ] |
51-75% (< 18 hrs) [ 3 ] |
>75% (< 24 hrs) [ 4 ] |
|
| Forecasted/Actual Total Rainfall, weekly between V4 and R4 stages | ||||
< 0.1 in [ 1 ] |
0.1-0.5 in [ 2 ] |
0.5-1.0 in [ 3 ] |
> 1.0 in [ 4 ] |
|
| Forecasted/Actual Daily High Temperature, weekly average between V4 and R4 stages | ||||
< 64°F [ 1 ] |
64-70°F [ 2 ] |
71-82°C [ 3 ] |
< 82°F [ 4 ] |
|
Common Bean Crop Rotation and/or Other Susceptible Hosts Included in Rotation |
||||
3 or more years [ 1 ] |
Less than 3 years [ 2 ] |
|||
| Susceptible Hosts and/or Fungal Apothecia Observed Nearby (less than 2 km) before R1 Stage | ||||
No Hosts, No Apothecia Nearby [ 1 ] |
Hosts OR Apothecia Nearby [ 2 ] |
Hosts AND Apothecia Nearby [ 4 ] |
||
| Timing AND/OR Amount of Nitrogen Fertilizer up to the Growth Stage noted; assume maximum rate of 112-168 kg/ha applied + carryover = 1X amount | ||||
Planting and < 1X [ 1 ] |
Planting and > 1X [ 2 ] |
V4 and > 1X [ 3 ] |
R1 and > 1X [ 4 ] |
|
| Total Water Applied/Received, weekly between V4 and R4 stages | ||||
< 1.4 in [ 1 ] |
–1.8 in [ 2 ] |
1.8-2.2 in [ 3 ] |
> 2.2 in [ 4 ] |
|
| Plant Spacing, Effects on Canopy Density, and Microclimatic Conditions Favoring White Mold | ||||
Wide rows, low to moderate density [ 1 ] |
Wide rows, moderate to high density [ 2 ] |
Narrow rows, moderate density [ 3 ] |
Narrow rows, high density [ 4 ] |
|
| Varietal Reaction (based on upright growth habit and/or genetic resistance) to White Mold | ||||
Resistant [ 1 ] |
Unknown [ 2 ] |
Susceptible [ 4 ] |
||
Bean Disease Risk — Total Score: |
||||
If your Total Score is:
|
| If the susceptible variety is planted in a field with a history of disease and the total score is 20 or higher, treatment with a labeled fungicide at 50-100% Bloom (R1 — every plant has one or more open blossoms) should be considered. A second application 7 days later at Full Bloom (7 to 10 days after R1) may be beneficial if conditions conducive to white mold continue. Pesticide information available at: http://www.highplainsipm.org/ |
This publication has been peer reviewed.
DisclaimerReference to commercial products or trade names is made with the understanding that no discrimination is intended of those not mentioned and no endorsement by University of Nebraska–Lincoln Extension is implied for those mentioned. |
Visit the University of Nebraska–Lincoln Extension Publications website for more publications.
Index: Plant Diseases
Dry Bean
2007, Revised October 2013