EC3068


Michael P. Carlson, Ph.D., Emeritus Assistant Professor of Practice
Connor Biehler, M.S., Beef Assistant Extension Educator
|
Mycotoxin |
Feedstuffs |
Mold(s) producing the mycotoxin |
Remarks |
|---|---|---|---|
|
Aflatoxins |
Corn, cottonseed, peanuts, sorghum, wheat, rice, and products derived from them |
Aspergillus spp., particularly A. flavus, A. parasiticus, & A. nomius |
|
|
Ergot alkaloids |
Cereal grains: rye, wheat, barley, and oats; triticale; tall fescue grass, and other grasses (e.g. brome) |
Claviceps purpurea (ergot toxicosis, ergotism); Neotyphodium coenophialum (fescue poisoning) |
|
|
Fumonisins |
Corn, barley, millet, oats, wheat, and products derived from them |
Primarily Fusarium verticilloides (formerly F. moniliforme), F. proliferatum |
|
|
Deoxynivalenol (DON, vomitoxin) |
Wheat, barley, triticale, other grains |
Fusarium spp |
Often found in grain of plants suffering from Fusarium head blight; often occurs with zearalenone |
|
Zearalenone |
Cereal grains, especially wheat, barley, and triticale |
Fusarium spp |
Often occurs with DON |
The purpose of this Extension Circular is to provide information to animal owners about the toxicology of mycotoxins with an emphasis on mycotoxins commonly found in Nebraska. The animals that are the focus of this circular include beef and dairy cattle, swine, sheep, goats, poultry, and horses.
Mycotoxins are toxic chemicals produced by molds that can poison animals or humans ingesting them. There are more than 300 identified mycotoxins; Table 1 lists mycotoxins commonly found in animal feedstuffs in Nebraska, the feedstuffs in which they occur, and the molds that produce them.
Mold growth occurs in two phases. During the initial or primary phase, the mold mass increases, which requires nutrients and energy to synthesize the biochemicals needed for mold growth. The secondary phase occurs after a period of sustained growth and usually involves mold reproduction.
During the secondary phase, molds produce chemicals called secondary metabolites, including mycotoxins. Secondary metabolites are not necessary during primary phase mold growth, but they contribute to the survival of molds in their environment, and some seem to help promote mold reproduction. Some secondary mold metabolites benefit humankind, such as the antibiotic penicillin, and some do not, such as mycotoxins.
Major factors affecting mycotoxin production by molds are mold strain; substrate; environmental conditions, especially temperature and relative humidity; water activity; and pH.
Molds that make mycotoxins, called mycotoxigenic molds, infect plant tissues. Those strains have the components of the mycotoxin synthetic pathway encoded in their genes so they can make those components and subsequently make mycotoxins. Not all strains of a particular mold are mycotoxigenic. For example, the percentage of the A. flavus population capable of producing aflatoxins (aflatoxigenic) varies from 40% to > 70%.
Fig. 1
The substrate is the plant tissue in which the infecting mold grows. The plant tissue provides the nutrients for mold growth and reproduction. The infecting mold may be specific to a particular kind or part of a plant.
The susceptibility of the substrate to mold infection is an important factor. Crops bred to resist mold infection are more likely to be mycotoxin free. However, insect or mechanical damage to the substrate increases the risk of infection and consequential mycotoxin production.
The presence of visible mold growth on the substrate does not necessarily indicate the presence of mycotoxins, nor does the absence of visible mold growth indicate the absence of mycotoxins. Chemical analysis is the best way to detect mycotoxin contamination in feeds. See Extension Circular Sampling and Testing Feed for Mycotoxins, EC3069 for more information about such analyses.
Mycotoxin production varies by climatic area. Each mycotoxigenic mold has optimal environmental conditions in which it germinates, grows, and produces its mycotoxin. Consequently, no single range of environmental conditions promote the production of mycotoxins.
Sometimes the relationship between environmental conditions and mycotoxin production is counterintuitive. Mold growth is generally associated with damp and warm conditions. However, aflatoxin contamination in dry-land corn infected by Aspergillus flavus increases during periods of drought and heat stress that occur during the reproductive stages of the corn plant.
Expect changes in mycotoxin production as weather patterns change over time. Changes in temperature extremes, precipitation patterns, insect infestations, and plant disease prevalence will affect mycotoxin production. Mycotoxin production changes that occur will likely be regional and could be advantageous or detrimental, depending on the climatic changes in the region. The scientific literature contains many papers discussing the impact of climate change on mycotoxin production; however, scientists lack the data that indicate what changes are occurring.
Mold growth occurs over a wide range of temperatures, reportedly between 5 oC and 35 oC (41 oF and 95 oF). Temperature ranges for optimal mold growth and mycotoxin production depend upon the mold that produces a specific mycotoxin, but generally, production is favored when temperatures are between 25 oC and 30 oC (77 oF and 86 oF).
Mycotoxin production is favored when the relative humidity is between 88% and 95%.
Periods of high humidity and warming temperatures at night in the spring of the year are very conducive to mold infection of plants and subsequent mold growth and mycotoxin production.
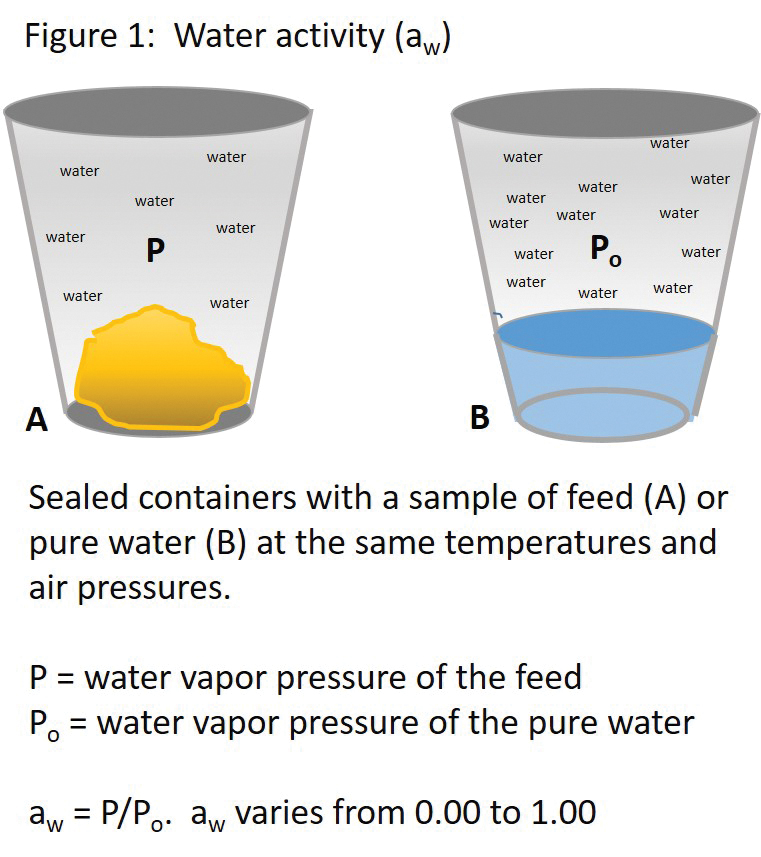
Water contained in food and feed exist in two pools; in this discussion about water activity, the word “feed” will be used in place of “food and feed.” One pool consists of water bound by various molecular interactions to chemicals in the feed and the other pool consists of free or unbound water. Water activity indicates the amount of “free water” in the feed and is the ratio of the water vapor pressure of the feed to the water vapor pressure of pure water (see Figure 1). The technicalities of water vapor pressure measurements are outside the scope of this document. Water activity values range between 0.00 and 1.00. A water activity of 0.00 means all of the water in feed is bound; a value of 1.00 means all of the water in the feed is free. Water activities at those extremes are very rare, if they ever occur.
|
Mycotoxin |
Mycotoxicosis produced |
Target tissue |
General description of clinical mycotoxicosis |
|---|---|---|---|
|
Aflatoxin |
Aflatoxicosis |
Liver |
Acute exposure produces liver failure; chronic exposure can produce liver cancer |
|
Ergot alkaloids |
Ergot toxicosis, ergot poisoning |
Central nervous system |
Nervous behaviors, convulsions |
|
Blood vessels |
Chronic exposure—tissue death and sloughing of tail and limbs |
||
|
Reproductive system |
Reproductive failure, reduced or absent milk production post-partum |
||
|
Summer slump, fescue toxicosis |
Thermal regulatory system |
Elevated body temperatures (hyperthermia) |
|
|
Fumonisins |
Porcine pulmonary edema (PPE) |
Lungs (swine) |
Fluid retention in lungs (pulmonary edema) |
|
Equine leukoencephalomalacia (ELEM) |
Brain; liver (horses) |
Neurological impairment; liver failure |
|
|
DON (vomitoxin) |
DON toxicosis, vomitoxicosis |
Gastrointestinal system, brain centers that control vomiting |
Feed refusal (anorexia), vomiting |
|
Zearalenone |
Zearalenone toxicosis |
Reproductive system, especially swine |
Females: disruption of estrus, pseudo-pregnancy. Young males: lower semen quality |
Microbes, including molds, use free water to germinate and grow, therefore, water activity of the feed is a better predictor of mold germination and growth than is feed moisture content. Generally, feed water activity greater than 0.7 favors mold growth. For example, Aspergillus flavus reportedly grows in substrates with water activity of at least 0.78 and aflatoxin production occurs with water activity of 0.84 to 0.87.
Water activities of animal feed are not readily available. Practically, feed moisture content is a crude predictor of water activity because the higher the moisture content, the higher the water activity. However, two feeds with the same moisture content may have very different water activities, making one susceptible to mold growth and the other less susceptible. That is because the relationships between moisture content and water activity differ from one feed to another. An example of differing water activities and water content for two human foods is salami and cooked beef. Both of those foods have approximately 60% moisture content, but the water activity of salami is about 0.82 and that of cooked beef is about 0.98.
pH is a measure of the acidity of a liquid or solution and ranges from 0 (extremely acidic) to 14 (extremely basic). The effect of the acidity of the environment in which the mold grows is complex and not completely understood. Mold cells can control local pH by secreting chemicals, but that capacity is limited. Acidic conditions tend to promote mold germination and mycotoxin production. Additionally, the stability of mycotoxin molecules varies with pH and they may disappear in very basic conditions.
Mycotoxins cause diseases called mycotoxicoses (plural for mycotoxicosis). Mycotoxins are absorbed into the body of the victim after the victim eats mycotoxin-contaminated feed and when enough of a mycotoxin is absorbed, disease occurs. Disease results when absorbed mycotoxin molecules, or their metabolites produced in the body, interact with target molecules on or in cells of the victim’s body. The disease produced depends upon how the mycotoxin alters the function of the target molecule.
Different mycotoxins cause different mycotoxicoses because mycotoxins or their metabolites interact with different tissues in the body. Table 2 lists examples of the mycotoxicoses produced by mycotoxins included in this document, the name of the mycotoxicosis each produces, their target tissue(s), and a general description of the mycotoxicoses produced.
Mycotoxicoses may be one of two types, acute or chronic. Acute mycotoxicoses occur after consuming highly contaminated feed once or a few times within a short time, usually a few hours. Chronic mycotoxicoses may occur after consuming less contaminated feed many times over a longer time, i.e., days, weeks, or months. Mycotoxin concentrations necessary to produce acute or chronic mycotoxicoses vary by mycotoxin and the animal species consuming the contaminated feed.
|
Characteristic |
Mycotoxicosis |
Mycosis |
|---|---|---|
|
Type and cause of the disease |
A poisoning caused by an ingested mycotoxin produced by a mold. The mold is not directly involved in the disease. |
An infection caused by a mold that invades a body tissue of the victim. The mold directly causes the disease. |
|
Antibody production by the immune system |
Usually none |
Yes |
|
Examples |
Aflatoxicosis |
Aspergillosis |
Mycotoxicoses are sometimes confused with mycoses, but they are very different diseases. Table 3 lists the characteristics of those two types of diseases.
The diagnosis of mycotoxicoses is not easy because other diseases or poisons may produce the same or similar clinical signs.1 A veterinarian must eliminate those other diseases or poisons to diagnose a mycotoxicosis. The veterinarian will usually use a process like that outlined below to diagnosis the cause of an animal health problem.
Results of various clinical diagnostic tests run on blood, serum, plasma, or urine specimens can indicate organ damage and help identify causes to include in the differential diagnosis. Staff at the veterinary clinic or at referral laboratories conduct such tests.
Observations made during microscopic examination of tissues may add, retain, or eliminate causes included in the differential diagnosis. Veterinary pathologists at veterinary diagnostic centers perform such examinations.
Chemical tests to detect the presence of mycotoxins in feed, which are readily available at commercial, veterinary diagnostic, and governmental laboratories, may be run. The presence of any mycotoxin in feed does not automatically confirm a mycotoxicosis; it does support the diagnosis of a mycotoxicosis. Ideally, the mycotoxin detected should be at a concentration sufficient to produce the illness experienced by the victims.
It may be challenging to collect a feed specimen that represents what the affected animals ate during exposure. Uneven distribution of mycotoxins in feed makes it possible to miss contaminated areas when a feed specimen is collected for analysis. Delayed specimen collection can miss contamination if the contaminated feed is gone by the time the specimen is collected. See the Extension Circular Sampling and Testing Feed for Mycotoxins, EC3069 for more information.
Chemical tests to detect the presence of mycotoxins in stomach or rumen content specimens may be run, if available at a testing laboratory. The presence of any mycotoxin in such specimens does not automatically confirm a mycotoxicosis, but it does support the diagnosis because it indicates exposure occurred.
Chemical tests to detect the presence of mycotoxins in tissue specimens are rarely conducted for two reasons. First, such tests are not readily available at testing laboratories. Second, interpretation of the results of those tests is challenging. Presence of a mycotoxin in a tissue specimen indicates exposure occurred, but it is difficult to associate tissue levels with the presence or absence of clinical signs or tissue effects.
The following evidence supports the diagnosis of a mycotoxicosis.
Once a mycotoxicosis has been diagnosed, the attending veterinarian can establish a treatment regimen.
Generally, there are no antidotes2 available to cure a mycotoxicosis. Treatment consists of five components.
We do not recommend that animal owners use the information provided in this section to diagnose or treat cases of suspected mycotoxicoses without the aid of a veterinarian because other poisons or diseases may cause the health effects and clinical signs listed for the mycotoxins. See the section of this document titled “Diagnosis of mycotoxicoses” for general information about mycotoxicosis diagnosis and treatment.
Aflatoxins produced by molds in the Aspergillus genus cause aflatoxicoses. Aflatoxins are poisonous to all classes of animals.
Aflatoxins may contaminate corn, milo, and cottonseed. Corn silage made from drought-stressed corn may be contaminated with aflatoxin. Distiller’s grain may also be contaminated. Historically, aflatoxin concentrations in feeds produced in Nebraska seldom are high enough to cause an aflatoxicosis. However, the frequency and amounts of aflatoxins found in corn grown during periods of drought increase.
There are five important aflatoxins named aflatoxin B1, B2, G1, G2, and M1. Aflatoxin M1 (AFM1) is a metabolite produced in the body after aflatoxin B1 (AFB1) is absorbed into the blood stream. Aflatoxins B1 and M1 can cause liver cancer.
Milk produced by lactating females after ingesting AFB1 may contain AFM1, so nursing animals may be exposed. Dairy products made from contaminated milk may be contaminated.
U.S. federal regulatory authority limits aflatoxin concentrations in commercially available feed and dairy products to protect animals and humans from excessive aflatoxin exposure. See the Extension Circular Use of Mycotoxin-Contaminated Feed for Animals, EC3066 for additional information about regulatory limits for aflatoxin-contaminated feed and dairy products.
Aflatoxins are toxic to companion animals, livestock, poultry, aquatic animals, and humans. Exposure is usually by eating aflatoxin-contaminated feed or food.
They are potent liver poisons. Acute exposure can cause liver failure; chronic exposure can cause liver cancer, which is of special concern for humans. Other effects attributed to aflatoxin exposure include immune system suppression (immunocompromise), birth defects, reproductive dysfunction, and reduced growth.
Amounts and durations of exposure affect the clinical signs produced by aflatoxins.
Acute exposure to lethal amounts may cause loss of appetite (anorexia), depression, weakness, prostration, difficulty breathing (dyspnea), vomiting (emesis), diarrhea, low body temperature (hypothermia) or fever, and death. Yellow discoloration of mucous membranes or cornea of the eyes (jaundice) may occur, which indicates significant liver damage.
Chronic exposure to sublethal amounts often produce more subtle changes. The first signs may be reduced weight gain and rough hair coats. Eventually liver damage may lead to anemia, jaundice, anorexia, and depression. The incidence of and time-to-recover from common infectious diseases may increase. It may also decrease the effectiveness of vaccines.

No antidote exists for aflatoxicosis.
Fig. 2
Promptly stop exposure by removing contaminated feed and replace it with nutritious, uncontaminated feed.
Activated charcoal administered to exposed individuals may help minimize absorption of aflatoxin already ingested and still present in the digestive tract.
Treat individual sick animals symptomatically and supportively.
Recovery of animals treated for liver damage may take some time, especially animals with extensive liver damage; such animals may never fully recover. Periodic assessment of liver function of ill animals may help determine the rate and extent of recovery.
Adjust vaccination and infectious disease treatments as necessary for immunocompromised animals. Strengthen biosecurity measures against infectious diseases as necessary.
It is best to prevent any exposure, especially for dairy animals. Use feed known to be aflatoxin-free. Be especially vigilant when using or purchasing corn grown during times of drought. The FDA has established acceptable limits for aflatoxins in feeds and milk. It also regulates blending contaminated feed with uncontaminated feed to reduce aflatoxin concentrations. See Use of Mycotoxin-Contaminated Feed for Animals, EC3066 for more information.
Mold-retardants applied to feed may help reduce the risk of aflatoxin-production in silage or harvested grain. Ammoniation of corn or cottonseed may reduce aflatoxin already present in those commodities; see the Extension Circular Use of Mycotoxin-Contaminated Feed for Animals, EC3066 for more information.
Ergot alkaloids (EAs) produced by Claviceps molds cause ergot alkaloid toxicoses. Ergot alkaloids may contaminate cereal grains such as rye, wheat, barley, triticale, and oats; and grasses such as brome and fescue.
Ergot alkaloid toxicoses have various names, some that indicate the exposure source, like ergotism, fescue foot, and fescue toxicosis. Other names are based on the general adverse effects or time of occurrence, like summer slump, summer syndrome, and fat necrosis. Ergotism applies to adverse effects occurring after the ingestion of sclerotia produced by Claviceps species of molds. Fescue foot and fescue toxicosis apply to adverse effects that occur after ingestion of fescue grass or hay.
Adverse effects produced depend upon the predominant ergot alkaloids present in the contaminated feed, the amount of contaminated feed ingested, duration of exposure, and animal species exposed. The scientific literature contains reports of EA toxicoses in humans, beef and dairy cattle, sheep, horses, and avian species. Ergotism and fescue toxicosis share several adverse effects.
Treatment of all forms of EA toxicoses are similar and listed at the end of this section.
Ergot is the name given the fungal infection of cereal grain and grasses by molds of the genus Claviceps. The presence of sclerotia (ergot bodies) in the seed heads of the infected plant is characteristic of the disease. Sclerotia bodies replace some of the seeds in infected seed heads.
Sclerotia are hard, dark-colored masses visible to the naked eye. Their shapes vary; some look like claws or talons and others look like rodent droppings. Their sizes range from a fraction of an inch to inches in length. Figure 2 illustrates sclerotia in seed heads of fescue grass.
Sclerotia contain EAs; which ones and their amounts present will vary. Chemical analysis can determine EA content. See Sampling and Testing Feed for Mycotoxins, EC3069 for more information about ergot alkaloid analyses.
Federal U.S. regulations limit the amount of sclerotia allowed in grains marketed commercially. See Use of Mycotoxin-Contaminated Feed for Animals, EC3066 for more information.
Exposure to EAs is associated with many adverse effects, which occur when EAs interact with cells in various parts of the body.
One way of classifying clinical ergotism is based on clinical effects: gangrenous, hyperthermia/production loss, reproductive failure, and convulsive/nervous.
Gangrene is tissue death (necrosis) due to loss of blood supply to the tissue. Some ergot alkaloids cause small blood vessels in the limbs, tips of the ears, and end of the tail to constrict reducing blood flow in those tissues, which can produce gangrene. The wattle, comb, beak, and feet in birds may be affected. This form generally occurs sporadically after several days to weeks of exposure.
Clinical signs of gangrenous form. Swelling and tenderness in the limbs, lameness; swelling of ears, or tail. Affected tissue becomes cold, dry, and discolored. The necrotic toes, ear tips, or tail may slough off.
Beef and dairy cattle are particularly affected. This form occurs when EAs disrupt the ability of the body to regulate its temperature causing the body to overheat (hyperthermia). Disruption can occur when the temperature regulation center in the central nervous system is affected and when vasoconstriction of peripheral blood vessels interferes with heat dissipation. Extreme hot and cold temperatures and high humidity exacerbates this type.
Hyperthermia is sometimes associated with production loss.
Clinical signs of hyperthermia/production loss. Heat intolerance; increased body temperature, heart and breathing rates; difficulty breathing; reduced feed intake and weight gain, weight loss; milk production reduction; poor body and hair coat conditions. Ergot alkaloid contamination of milk has not been reported.
Dairy cows and mares are particularly affected. This form occurs when EAs interact with various cells associated with reproduction.
Clinical signs of reproduction loss. Infertility/reproductive failure, prolonged gestation period, malformed offspring, abortion, still births, and reduced or absent milk production at or after parturition.
Reports of this form of ergotism in animals are rare and not well documented. It requires the acute ingestion of very high doses of EAs. Governmental regulations that limit the amount of sclerotia in cereal grains protect against the occurrence of this form.
This form occurs when EAs interact with various parts of the brain.
Clinical signs of convulsive/nervous form. Central nervous system abnormalities such as incoordination or staggering gait (ataxia) and confusion.
The name “fescue toxicosis” arose when adverse health effects were closely associated with ingestion of tall fescue grass (Festuca arundinacea Shreber). Fescue grass does not actually cause the toxicosis; EAs produced by the mold Epichloë coenophiala (formerly Neotyphodium coenophialum) do. That mold is present as an endophyte in tall fescue. An endophytic mold lives between a plant’s cells in a symbiotic relationship where the plant provides the mold with nutrients and the mold benefits the plant. This mold increases the survivability of fescue grass under unfavorable growing conditions, like drought and heavy grazing.
Fescue toxicoses have other names, including fescue foot, summer slump, summer syndrome, summer fescue toxicosis, and fat necrosis. Cases most commonly occur in cattle, but can occur in other species, too.
Fescue grasses are susceptible to infections by Claviceps species of mold, too. The presence of sclerotia in the fescue seed heads poses a risk of ergotism. Theoretically, both ergotism and fescue toxicosis could occur concurrently in endophyte-containing fescue varieties, but documentation of such occurrences is lacking.
Clinical signs associated with fescue toxicosis are similar or the same as those associated with ergotism described above. Importantly, the clinical signs correlate with the ingestion of fescue.
Fescue foot is indistinguishable from gangrene of the extremities associated with ergotism. It occurs commonly in late fall or winter. Endophyte EAs can increase the risk of painful inflammation of the hoof, which can result in life-threatening lameness. Clinical signs appear from one to several weeks after commencement of fescue grass feeding and commonly occur in one or both hind limbs.
Summer slump/summer syndrome/summer fescue toxicosis is a common and economically important problem in cattle associated with grazing fescue pastures or eating fescue hay. It is like the hyperthermia/production loss form of ergotism described above and becomes evident during summer heat spells. It may also include the reproductive problems listed above, which may be important to cattle owners who calve during the fall.
Pregnant mares are most susceptible to fescue toxicosis after 300 days of gestation (gestation length of 335—345 days). Stop exposure to endophyte-infected fescue during last month of gestation because continued exposure may cause prolongation of gestation by about a month, prevent lactation, and produce “fescue foals” or “dummy foals.” Fescue foals may be smaller or larger than normal; larger foals increase the risk of difficult birth with trauma to the uterus, cervix, or vagina. Mares with damage to their reproductive tract may experience rebreeding problems. The placenta may detach, and its expulsion may precede that of the foal presenting as a red bag. Such occurrences threaten the life of the foal and mare. Retention of fetal membranes is more likely.
Fat necrosis occurs in many ruminant species and is associated with the presence of necrotic fat in abdominal and pelvic cavities. The presence of such masses may be asymptomatic, but their presence may become important if associated with gastrointestinal effects, like chronic bloat, colonic constriction, or obstruction. A veterinarian must diagnose it.
Sclerotia may be found in specimens of the diet, dietary components, or stomach/rumen contents. Feed specimens or sclerotia found in feed may be analyzed for EA content. Detection of sclerotia or EAs in feed specimens indicates exposure may have occurred; detection in stomach/rumen content indicates exposure has occurred.
Analyses for EA content of blood, serum, plasma, urine, or intestinal content are not usually available at diagnostic laboratories.
Prolactin analysis of serum or plasma may be conducted in cases of reduced or absent milk production postpartum.
Identification of a Claviceps or endophyte mold presence in plant specimens by culturing is not usually done.
Early recognition of signs and rapid diagnosis help reduce the duration and severity of the toxicoses.
Remove the source of exposure and feed an uncontaminated, nutritious diet.
Provide supportive treatment until clinical signs abate. Restoration of gangrenous limbs, ears, or tails is unlikely. Severely damaged limbs may necessitate euthanasia of affected animals.
Some drugs may be appropriate for treatment of certain effects of ergot alkaloids and limited to certain species. Consult a veterinarian for more information.
Prevent re-exposure by using grains known to be free of sclerotia or ergot alkaloid contamination.
Contaminated grain may be cleaned of sclerotia by mechanical means. The rejected portion containing the sclerotia should never be used as feed because the amount of sclerotia on a percentage basis in it is likely greater that in the original grain.
Graze or feed fescue varieties that do not contain the endophytic mold.
Fumonisins, produced by Fusarium moniliforme and F. proliferatum, cause this type of mycotoxicosis. They have been detected in barley, millet, oats, and wheat, but by far, the most important source of animal exposure is contaminated white or yellow corn or products derived from them.
The most prevalent of the fumonisins are fumonisin B1 (FB1), fumonisin B2 (FB2), and fumonisin B3 (FB3); the most important of those three is FB1.
Fumonisins act by disrupting the production of a biochemical class of compounds called sphingolipids. Sphingolipids are important components of cell membranes that help maintain membrane structural integrity and they play significant roles in cell growth, tumor production, inter-cellular communication, and cellular functional development.
Organs damaged by fumonisin include the liver, lungs, brain, kidney, esophagus, heart, and immune system. Most important to veterinary medicine are damage to the lungs of pigs (porcine pulmonary edema, PPE) and the brains of horses (equine leukoencephalomalacia, ELEM).
Cattle are relatively resistant to toxic effects of fumonisin.
There is experimental evidence that poultry exposed to fumonisin may develop diarrhea, experience decreased body weight gain, and experience liver damage. An acute death syndrome called spiking mortality syndrome in young chicks, originally thought to be related to fumonisin exposure, has been associated with another Fusarium mycotoxin called moniliformin and not fumonisin.
This type of fumonisin toxicosis occurs in pigs. Fumonisin exposure results in fluid accumulation in the lungs (pulmonary edema); death occurs because that edema causes respiratory failure.
Fumonisins cause pulmonary edema indirectly by decreasing the ability of heart muscles to contract. That leads to a functional left-sided heart failure, which causes back-pressure within the pulmonary vasculature and results in pulmonary edema.
Clinical signs of respiratory distress usually develop 3 to 6 days after initial exposure and include difficulty breathing, open-mouth breathing, increased respiratory rate, discoloration of skin and mucous membranes (cyanosis), inactivity, and death within a few hours of onset of signs of respiratory distress.
Fumonisin exposure causes degeneration and death of the white matter in one or both sides of the brain of horses. Damaged areas can be visible to the naked eye at necropsy as cavitations in the brain. Liver damage may also occur concurrently. Death can result from damage to either the brain or liver. Some animals will show signs of damage to both organs.
Those indicating brain damage: sudden onset of aimless circling, blindness, head pressing, partial paralysis, incoordination, depression or hyperexcitability/frenzy. There are some reports of difficult and labored breathing.
Those indicating liver damage: yellow discoloration (jaundice/icterus) of mucous membranes or whites of the eye (sclera); small red or purple spots (petechiae) on mucous membranes, swelling of lips or muzzle.
Serum specimens may be analyzed for evidence of liver dysfunction.
Ratios of certain kinds of sphingolipids present in blood may be determined.
Stomach contents and feed specimens may be analyzed for fumonisins. The presence of fumonisins in stomach content specimens indicates exposure and helps support the diagnosis.
Microscopic examinations of sections of brain, liver, and lung tissue specimens may be conducted. The presence of lesions caused by fumonisin helps support the diagnosis.
Culturing of feed to identify any Fusarium molds present in feed is generally not useful because feed may be highly contaminated with fumonisins in the absence of visible mold growth, or feed heavily infected with Fusarium mold may not have detectable amounts of fumonisins in it.
Remove the contaminated feed immediately and replace it with a nutritious, uncontaminated diet.
There are no antidotes for either ELEM or PPE. The progression of the disease to death after onset of clinical signs is usually rapid for both types.
Prevent exposure or re-exposure by identifying and rejecting fumonisin-contaminated feed or feed stuffs. Use feed known to be free of fumonisin contamination. The U.S. FDA has established guidelines for maximum fumonisin content in feeds for use by various animals. See Use of Mycotoxin-Contaminated Feed for Animals, EC3066 for more information.
The U.S. FDA approved fumonisin esterase as an additive to degrade fumonisins present in swine and poultry feed. See Use of Mycotoxin-Contaminated Feed for Animals, EC3066 for more information.
Detection of fumonisins in feed requires chemical analysis. See Sampling and Testing Feed for Mycotoxins, EC3069 for more information.
Feeding corn screenings to any animal is very dangerous, especially for horses. Screenings may contain very high concentrations of fumonisins. Screenings are often cheaper than better quality corn, but they can kill pigs or horses consuming them.
Vomitoxin is a member of a group of mycotoxins called tricothecenes. Other tricothecenes are T-2 toxin, HT-2 toxin, diacetoxyscirpenol (DAS), and nivalenol. Vomitoxin is the most important of those mycotoxins in the United States and Canada.
Fusarium species of mold produce tricothecenes. Those molds cause disease in plants, including head blight in wheat, barley, triticale, and other grains In North America. Vomitoxin often occurs with another Fusarium mycotoxin called zearalenone. Zearalenone is discussed later in this document.
Vomitoxin exposure is not deadly, but it can affect the productivity of animals by adversely affecting the gastrointestinal tract, which can alter absorption of nutrients. That can lead to reduced feed efficiency and subsequently reduce weight gain. Sensitivity to the effects varies among species. Ruminants, horses, and poultry are not very sensitive; swine and dogs are.
There is evidence that vomitoxin may affect functions of the immune system, but the evidence is not consistent.
Vomitoxin is relatively stable and does not degrade when contaminated grain is milled or processed into feed. It is stable at temperatures up to 120 oC (248 oF).
In pigs, chronic exposure to relatively low doses produces feed refusal, weight loss, impaired performance, and weakness. Excessive, acute exposure to higher doses produces signs of gastrointestinal distress (abdominal pain, diarrhea), vomiting, and feed refusal. Excessive, acute exposure may also increase susceptibility to infection and decrease the effectiveness of vaccinations.
Samples of feed and stomach content may be analyzed for vomitoxin. Detection of its presence in those samples supports a diagnosis of vomitoxicosis.
Vomitoxicosis is rarely, if ever, fatal. No antidote exists to treat vomitoxicoses.
Remove the source of exposure and replace it with a nutritious, uncontaminated diet.
Treat the symptoms and support the affected animals until they recover.
Prevent recurrence by using feed known to be vomitoxin free. The U.S. FDA has established advisory levels for vomitoxin in animal feeds. See Use of Mycotoxin-Contaminated Feed for Animals, EC3066.
Zearalenone, produced by molds in the genus Fusarium, contaminates wheat, sorghum, barley, and oats. Most importantly, it can contaminate corn and products derived from corn, including corn silage. It rarely contaminates forages. It was first called F-2 toxin and often occurs concurrently with vomitoxin. It can survive the heat of pelleting and is stable in the environment, so it can persist for a long time.
Zearalenone acts as an estrogenic compound, even though its chemical structure is not like estrogen, by binding to estrogen receptors causing estrogenic effects. Swine are the most sensitive to its effects, especially gilts and young boars.
Ruminants are not very sensitive, but there are reports of effects in dairy heifers, cows, and ewes. Zeranol (α-zearalanol), a derivative of zearalenone and marketed as RALGRO®, is a growth-promoter approved for use in cattle and sheep production.
Poultry are resistant to its effects.
Field cases of zearalenone exposures in swine report swollen vulva and mammary gland swelling, especially in prepubertal gilts. It can disrupt the estrus cycle of female pigs. Boars may have reduced libido.
There are reports of reduced conception rates in cattle. Young dairy heifers may exhibit enlarged mammary gland in one or more quarters, turbid discharge from the vulva, or estrous behavior. Decreased milk production or infertility may occur in dairy cows and heifers.
Generally, estrogenic compounds like zearalenone are not abortifacients, so later-term abortions are not caused by zearalenone exposure. However, it could prevent a fertilized egg from implanting in the uterine wall.
Instances of feed refusal associated with zearalenone exposure in pigs are more likely to due to vomitoxin present concurrently with zearalenone.
Clinical signs observed during controlled feeding trials are more extensive because the animals are observed more closely, and effects measured using diagnostic tests not commonly run for field cases. Sometimes the animals were dosed with purified zearalenone. How such studies apply to field cases is debatable. Signs reported from such studies are summarized below.
Pregnancy (females) or reproductive fitness tests (males or females) may be run.
Feed specimens or stomach/rumen content may be analyzed for the presence of zearalenone.
Stop exposure by removing zearalenone contaminated feeds. Replace it with a nutritious, uncontaminated diet. Clinical signs usually abate within days of cessation of exposure but may take longer after exposure to high concentrations. Full recovery is the norm.
Prevent recurrence by using feed known to be free of zearalenone contamination. The U.S. government has not regulated the amount of zearalenone that can be present in commercially available feed. See Use of Mycotoxin-Contaminated Feed for Animals, EC3066 for recommendations based on European Union regulations.
Mycotoxins are toxic chemicals produced by molds that infect plants and grow in feed. They are secondary metabolites produced by molds, which contribute to the survival or reproduction of the molds.
Five mycotoxins are commonly found in Nebraska: aflatoxins, ergot alkaloids, fumonisins, deoxynivalenol, and zearalenone.
Major factors affecting mycotoxin production include mold strain, substrate, environmental conditions, water activity and pH.
Diseases caused by mycotoxins, called mycotoxicoses, vary because mycotoxins differ in how they interact with and affect cells in the body.
The diagnosis of mycotoxicoses is not easy and requires the expertise of a veterinarian. Clinical signs indicative of a mycotoxicosis support the diagnosis, as do results of clinical and specialized diagnostic tests that indicate or confirm known effects of a mycotoxin. Elimination of other causes of illnesses similar to a mycotoxicosis is required. The presence of a mycotoxin in feed consumed by victims at concentrations high enough to cause the illness experienced by the victims supports the diagnosis of a mycotoxicosis.
There are no antidotes available to cure a mycotoxicosis. Treatment consists of stopping exposure; minimizing absorption of mycotoxins already consumed, treating the victim symptomatically and supportively, and preventing recurrence of the mycotoxicosis.
1. Clinical signs are indicators of illness or disease recognizable by an observer; examples of signs are a rash or vomiting. The phrase “clinical sign” is not synonymous with “symptom.” Symptoms are subjective indicators of illness or disease perceived and communicated by the victim. Veterinary patients cannot tell us the symptoms they may be experiencing. Signs may accompany symptoms, for example, a rash may itch.
2. Antidotes are drugs that cure a poisoning by interfering in some way with the interaction of the poison and its molecular target. The interference helps restore the health of the victim.
Alshannaq, A. & J. Yu (2017) Occurrence, Toxicity, and Analysis of Major Mycotoxins in Food, International Journal of Environmental Research and Public Health, Vol 14, No. 6, pages 632 and following.
Anonymous (2022) Food Additives Permitted in Feed and Drinking Water of Animals; Fumonisin Esterase in the Federal Register, Vol 87, No. 148, August 3, 2022, p. 47343.
Anonymous (2022) Food Additives Permitted in Feed and Drinking Water of Animals; Fumonisin Esterase, in the Federal Register, Vol 87, No. 166, August 29, 2022, p. 52681.
Anonymous, (1984) Water Activity (aw) in Foods, US FDA accessed during Aug 2022 at https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/inspection-technical-guides/water-activity-aw-foods#:~:text=The%20water%20activity%20(a%20w)%20of,distilled%20water%20under%20identical%20conditions.
Calvo, AM, et al (2002) Relationship between Secondary Metabolism and Fungal Development, Microbiology and Molecular Biology Reviews, Vol 66, No. 3, pages 447–45.
Daou, R, et al (2021) Mycotoxins: Factor influencing production and control strategies, AIMS Agriculture and Food, Vol 6, No. 1, pages 416–447.
Ferguson, TD, ES Vanzant, & KR McLeod (2021) Endophyte Infected Tall Fescue: Plant Symbiosis to Animal Toxicosis, Frontiers of Veterinary Science, vol 8, page 774287. Doi:10.3389/fvets.2021.774287.
Gupta, R.C. (2018), Section XV: Mycotoxins in Veterinary Toxicology Basic and Clinical Principles, Third Edition, Academic Press:
Chapter 69—Aflatoxins, by Coppock, Christian, & Johnson;
Chapter 70—Ergot and Fescue Toxicosis, by Gupta, Evans, & Nicholson;
Chapter 71—Fumonisins, by Smith;
Chapter 75—Tricothecenes, by Cope;
Chapter 76—Zearalenone, by Gupta, Mostrom, & Evans.
Hafen BB, Burns B. (Updated 2021 Aug 30) Physiology, Smooth Muscle, in StatPearls [Internet], Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK526125/
Medina, A., A. Rodriguez, & N. Magan (2016) Climate change and mycotoxigenic fungi: impacts on mycotoxin production, Current Opinion in Food Science, vol 5, pages 95–104.
Morgavi, D.P. & R.T Riley (2007) “An (sic) historical overview of field disease outbreaks known or suspected to be caused by consumption of feeds contaminated with Fusarium toxins,” Animal Feed Science and Technology, vol 137, pages 201–212
Mostrom, Michelle S (2021) “Ergotism in Animals,” Merck Veterinary Manual Professional Version, accessed Sep 16, 2022 at https://www.merckvetmanual.com/toxicology/mycotoxicoses/ergotism-in-animals
Pleadin, J., et al (2019) Chapter 8—Mycotoxins in food and feed in Toldra, F. (Editor) Advances in Food and Nutrition Research, vol 89, pages 302 and following.
Shier, W.T. (2011) On the origin of antibiotics and mycotoxins, Toxin Reviews, vol 30, No. 1, pages 6–30.
Tapia, M.S., et al, Chapter 14—Effects of Water Activity (aw) on Microbial Stability as a Hurdle in Food Preservation, Barbosa-Canovas, G.V., et al (Editors) (2020) Water Activity in Foods Fundamentals and Applications, Second Edition, IFT Press & Wiley Blackwell.
This publication has been peer reviewed.
Nebraska Extension publications are available online at http://extensionpubs.unl.edu/.
Extension is a Division of the Institute of Agriculture and Natural Resources at the University of Nebraska–Lincoln cooperating with the Counties and the United States Department of Agriculture.
Nebraska Extension educational programs abide with the nondiscrimination policies of the University of Nebraska–Lincoln and the United States Department of Agriculture.
© 2023, The Board of Regents of the University of Nebraska on behalf of the Nebraska Extension. All rights reserved.